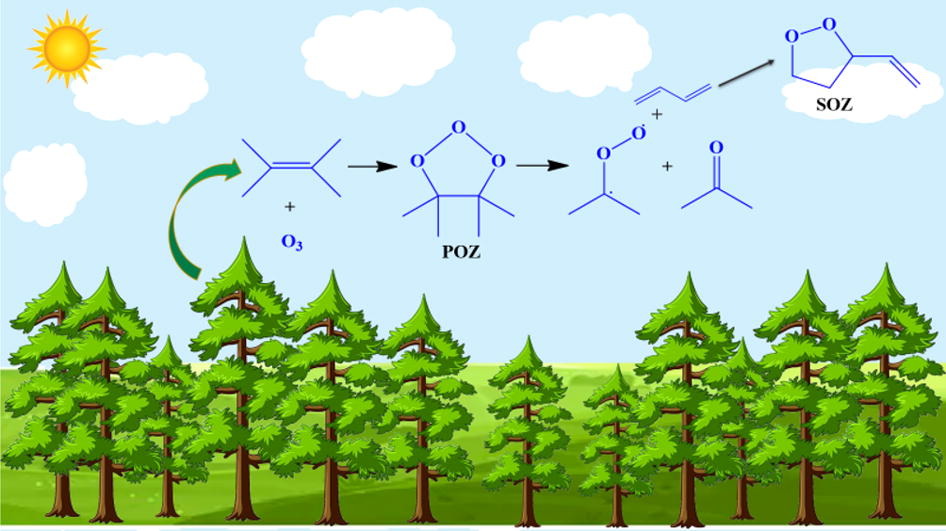
The reaction kinetics of 1,3‑butadiene with Criegee Intermediate (CH2OO, CI) was investigated over T = 200–400 K using the CCSD(T)/aug-cc-pVTZ//B3LYP/6-311G (d, p) level of theory. Reaction pathways involving distinguished conformational addition variants of both the reactants proceeds via a 1,3-dipolar cycloaddition to form the SOZ. The effect of temperature suggested that, the contribution by the inner-facing TSs decreases, while outer-facing reaction channels become more significant, as the temperature increases. At low pressures, the effect of pressure was more prominent at temperatures close to 200 K. However, at 760 Torr, the effect was insignificant over the complete temperature range.
-
Call
-
E-mail
Journal Details
1. Reaction kinetics of CH2OO with 1,3-butadiene: Mechanistic investigation with RRKM calculations.
P. Gupta & B. Rajakumar Chem. Phys. Lett, 2020, 742, 137157.